Legal cannabinoids and THC alternative medicines have been approved for medicinal use. Other medicines that mimic the properties of marijuana are currently undergoing clinical trials before FDA approval. The FDA can allow companies to conduct clinical trials of drugs that are intended to treat disorders, even if they’re marijuana-based drugs.
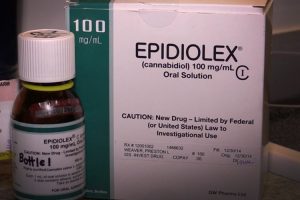
Epidiolex
Not yet fully approved by the FDA, Epidiolex is currently in its clinical trails. GW Pharmaceuticals has been developing and testing multiple cannabinoid drugs. It contains a highly purified, plant-derived form of cannabidoil (CBD), which is the non-psychoactive compound present in marijuana plants.
The “high” from ingesting marijuana comes from THC, the main psychoactive ingredient in THC. Epidiolex utilized CBD instead of THC. CBD has been known to treat Dravet syndrome, a rare and sever form of epilepsy in children. Epidiolex has been found to be beneficial in the treatment of Dravet and Lennox-Gastaut Syndrome, another uncommon form of childhood epilepsy.
Syndros
Syndros is just the generic version of Marinol that was recently approved by the FDA. The active ingredient in Syndros is also Dronabinol and the drug is being used to treat the same ailments as Marinol.
Both drugs have been found useful in treating anorexia associated with weight loss in patients suffering from AIDS. Patients going through chemotherapy hve found the drug to relieve their weight loss, nausea, and vomiting associated with the chemotherapy.
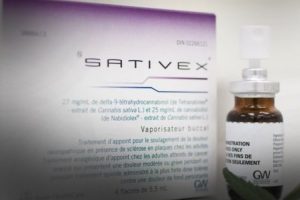
Sativex
Sativex is another drug not approved by the FDA currently undergoing clinical trials. GW Pharmaceuticals is also in charge of developing and testing Sativex. It is being used to treat spasticity due to multiple sclerosis in 25 countries.
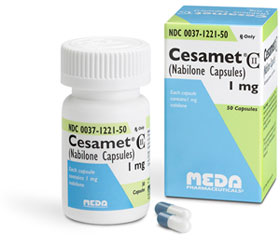
Cesamet
Cesamet is a FDA-approved medicine which is a schedule II drug that is only available through prescription. It has already undergone its clinical trials and has been found to help cancer patients dealing with loss of appetite, nausea, and vomiting. It is meant to be orally administered.
The active ingredient in Cesamet is called Nabilone, which is similar to the active ingredient in Cannabis, THC. The active synthetic cannabinoid nabilone has complex effects on the central nervous system. It interacts with the cannabinoid receptor system in the same way THC and other legal cannabinoids would.
Marinol
Marinol is a THC-based drug that received FDA approval in 1985. The synthetic form of THC has made a return in liquid form recently. Dronabinol is the active ingredient in Marinol. It is taken in capsule form and it is a light yellow resinous oil that is sticky at room temperatures and hardens upon refrigeration much like hash oil.
The Next Steps
Opponents of legalized marijuana believe there are enough FDA-approved alternatives to prevent the legalization of marijuana in its plant form. However, these opponents are disregarding the benefits of the entourage effect. The entourage effect provides additional medical benefits when the various components of marijuana combine. When cannabinoids and THC are separated, they may not be as helpful to certain patients. More research needs to be done on these legal alternatives if they are going to replace actual marijuana as medicine.